magicmine/iStock through Getty Pictures

Temporary Historical past Of Alpine To Current Day, Together with Latest Bull Run On Kidney Illness Information
Alpine Immune Sciences, Inc. (NASDAQ:ALPN), headquartered in Seattle, joined the Nasdaq in June 2017, through a merger with failing Cystic Fibrosis drug developer Nivalis Therapeutics.
Backed by a number of biotech enterprise capital corporations, together with Frazier Healthcare Companions, Alpine BioVentures, and OrbiMed Advisors, the mixed firm started life with ~$90m money, and a share worth of ~$10. At the moment, the corporate mentioned its enterprise as follows:
Alpine Immune Sciences, Inc. was based in 2015 and is targeted on creating novel protein-based immunotherapies utilizing its proprietary variant immunoglobulin area (vIgD™) platform expertise.
The vIgD platform is designed to work together with a number of targets, together with many current within the immune synapse. Alpine’s vIgDs are developed utilizing a singular course of referred to as directed evolution, which may produce proteins succesful of both enhancing or diminishing an immune response and thereby might apply therapeutically to each oncology and inflammatory ailments.
Alpine has additionally developed its transmembrane immunomodulatory protein (TIP™) expertise, based mostly on the vIgD platform, to boost engineered mobile therapies.
Alpine had established a worldwide analysis and growth program with Kite Pharma – now a part of Pharma big Gilead Sciences, Inc. (GILD), probably value as much as $535m in future milestone funds, plus royalties, nonetheless Kite walked away from the deal in 2019.
In the meantime, Alpine had been creating its lead candidate, ALPN-101, a “dual Inducible T cell Costimulator (“ICOS”), and CD28 antagonist intended for the treatment of autoimmune and inflammatory diseases,” and in 2020, entered right into a co-development partnership with the pharma big AbbVie, which included a $60m upfront cost and the choice for AbbVie to take over growth and industrial actions for the drug, if accredited, with >$800m of milestones probably on the desk for Alpine.
Ultimately, in December final yr, with Alpine’s Part 2 research of ALPN-101, now referred to as acazicolcept, overrunning, AbbVie gave Alpine the choice to terminate the research early, in alternate for a considerably decrease payout ought to AbbVie resolve to take up its possibility, with complete milestones falling to ~$150m most.
A second drug candidate developed by Alpine – davoceticept, or ALPN-2020, an investigational CD28 costimulator and twin checkpoint inhibitor, was shelved after two affected person deaths occurred throughout two scientific research, one evaluating the drug as a monotherapy, and a second alongside Merck’s mega-blockbuster checkpoint inhibitor keytruda, or pembrolizumab, in superior malignancies.
In brief, Alpine endured a tough begin to life as a public firm, its inventory plummeting to lows of <$4 in 2020, nonetheless, its saving grace has been a 3rd asset, povetacicept, or ALPN-303.
In its Q3 2023 quarterly report / 10Q submission, Alpine calls povetacicept “a dual antagonist of the B cell activating factor (“BAFF”), and a proliferation-inducing ligand (“APRIL”), cytokines, which play key roles in the activation, development, and survival of B cells.”
The 10Q goes on to state that:
povetacicept has exhibited larger efficiency in preclinical research versus wild-type TACI-based comparators, in addition to different inhibitors of BAFF and/or APRIL alone.
We’re evaluating povetacicept in our RUBY-3 and RUBY-4 research.
RUBY-3 is a a number of ascending dose, multi-cohort, open-label, section 1b/2a research of povetacicept in IgA nephropathy, lupus nephritis, and first membranous nephropathy, the place povetacicept is being administered subcutaneously as soon as each 4 weeks for as much as 48 weeks. Key endpoints embody proteinuria, eGFR, renal response, and disease-related autoantibodies.
RUBY-4 is a multi-cohort, open-label section 1b research of povetacicept in immune thrombocytopenia, autoimmune hemolytic anemia, and chilly agglutinin illness, the place povetacicept is being administered subcutaneously as soon as each 4 weeks for as much as 48 weeks. Key endpoints embody respective blood cell counts, together with sturdy responses, in addition to disease-related autoantibodies.
Since 2021, povetacicept’s preclinical information has attracted the eye of Wall Avenue analysts, serving to to maintain its share worth secure because the acazicolcept and davoceticept packages unravelled, and in November final yr, the corporate was in a position to share optimistic scientific information that has triggered a six-month bull run on shares, which have risen in worth by >215%, to commerce at $39 on the time of writing, valuing the corporate at ~$2.53bn.
Povetacicept’s Information – A Shot At Finest-in-Class Standing In Multi-Billion Kidney Illness Market?
Alpine selected to share its information in a late-breaking Poster Session on the American Society of Nephrology Kidney Week 2023, on Nov. 2, 2023. The headline information beneath is copied from an Alpine press release:
- As of Oct. 25, 20 members with IgA nephropathy (IgAN) have been enrolled, 12 on the 80 mg dose degree, of whom 5 have UPCR information out there at 24 weeks.
- In IgAN, remedy with low-dose povetacicept, 80 mg SC each 4 weeks was related to clinically significant enhancements in proteinuria, with a 53.5% discount from baseline in urine protein to creatinine ratio (UPCR; n=5) at 24 weeks. As well as, at 24 weeks, 4/5 (80%) had achieved remission, as outlined by UPCR < 0.5 g/g and ≥50% discount in UPCR from baseline with secure renal operate (≤ 25% discount in eGFR from baseline).
- In IgAN, remedy with low-dose povetacicept additionally was related to a >60% discount in the important thing disease-related biomarker galactose-deficient IgA1 (Gd-IgA1), in addition to secure renal operate as assessed by estimated glomerular filtration price (eGFR) (+7.1% from baseline at 24 weeks; n=5).
- The primary participant with major membranous nephropathy (pMN), additionally handled with povetacicept 80 mg SC each 4 weeks, achieved an immunological remission, outlined as a discount within the extremely disease-relevant biomarker anti-PLA2R1 to an undetectable degree, from a baseline of 209 to < 2 RU/mL by 22 weeks.
- Povetacicept has been effectively tolerated, with no reported administration-associated reactions, no situations of IgG < 3 g/L, and no extreme infections.
- The next dose of povetacicept, 240 mg SC each 4 weeks, continues to enroll, with preliminary information anticipated in 1H 2024.
IgAN is a illness characterised by hematuria (blood in urine), and proteinuria (excessive ranges of protein in urine), which may result in progressive renal (kidney) failure, and it impacts ~150k sufferers within the US, and even larger numbers in Europe and Asia.
IgAN has historically been handled with renin-angiotensin-aldosterone system (RAAS) blockade with immunosuppression, however not too long ago accredited medicine within the indication embody Calliditas Therapeutics AB (publ) (CALT) Tarpeyo, an oral, delayed-release formulation of the corticosteroid budesonide that acquired full approval in January this yr, having acquired accelerated approval in 2021, and Travere Therapeutics, Inc. (TVTX) Filspari, a non-immunosuppressive drug, awarded accelerated approval in February 2023 (accelerated approval permits the drug to be offered commercially, though the drug developer should conduct additional research, which, if unsuccessful, may see the drug withdrawn from the market).
In 2023, Filspari earned $29.2m of revenues in its first yr in the marketplace, whereas Tarpeyo earned ~$105m in revenues. Which will sound underwhelming, given Alpine’s $2.5bn market cap, nonetheless, though analysts have shared few ideas on the height income potential of povetacicept, Wall Avenue has speculated that Vera Therapeutics, Inc. (VERA) atacicept, which has a really comparable mechanism of motion to Alpine’s drug, being a self-administered fusion protein that blocks BAFF and APRIL, may obtain $1.25bn in peak gross sales within the IgAN indication alone, whereas Vera’s administration consider the annual market alternative for its drug is $6bn – $10bn.
I mentioned Vera in a deep dive note posted to Looking for Alpha shortly after the corporate’s inventory started rising on optimistic IgAN information for atacicept, in January this yr. Shares traded at $25 on the time, and I gave the inventory a “hold” advice, provided that approval of the drug relies on the end result of a separate Part 3 research and possibly 2-3 years away, however shares have continued to rise, to >$45 on the time of writing, valuing Vera’s enterprise at ~$2.5bn – the very same worth the market attaches to Alpine.
Vera’s information was from a Part 2b research, at 72 weeks, and was summarized in a press launch as follows:
Contributors handled with atacicept for 72 weeks demonstrated a 62% discount in Gd-IgA1, a discount within the proportion of members with hematuria to 19%, and a 48% discount in UPCR within the per-protocol (“PP”) evaluation.
Vera’s information appears to barely lag Alpine’s when it comes to UPCR – a 48% discount at 72 weeks, vs. 54% in simply 24 weeks – though it ought to be famous Alpine’s information is taken from simply 5 sufferers, and that in different areas, equivalent to estimated globular filtration price (“EGFR”), atacicept might have a slight edge, though the fact is we might not know for positive till we now have the total RUBY-3 information.
Povetacicept scientific expertise thus far (Alpine presentation)

As we will see above, a slide from a latest Alpine presentation suggests administration is assured that, even at this early stage of growth, povetacicept is exhibiting best-in-class potential, and administration expects to maneuver the drug right into a pivotal research in IgAN within the second half of this yr.
As such, it appears doable that povetacicept may nonetheless beat atacicept to market, if accredited, in fact, and if we think about that the list price of tarpeyo and filspari is ~$85k each year, and multiply this by the addressable affected person inhabitants of 150k folks within the US alone, we attain a determine of almost $13bn.
A ten% share of that market could be sufficient to make povetacicept a “blockbuster” (revenues of >$1bn each year), and contemplating industrial stage pharmas typically commerce at a price-to-sales ratio of 3-5x gross sales, it isn’t laborious to see why the share worth of Alpine has been climbing.
If administration can broaden the label of povetacicept into different indications, it may have a multi-billion promoting drug on its fingers, which means that buyers shopping for right this moment would have good purpose to consider the share worth and market cap valuation may double within the subsequent 3-5 years, to not point out the prospect of the corporate turning into a beautiful acquisition goal – supplied its drug works in addition to administration believes it can.
Dangers To The Alpine Bull Thesis – Intense Competitors, Poor Monitor Report, Funds
Earlier than getting too carried away with the best-case state of affairs, nonetheless, it is value contemplating some drawbacks to the bull thesis.
Kidney illness is a tough house for biotechs and drug builders characterised by excessive charges of failure, though latest approvals have arguably opened up the IgAN house considerably, and each povetacicept and atacicept are attaining ranges of UPCR discount – >40% – that the FDA might deem approvable, with apparently stable security profiles.
Nonetheless, there are different drug candidates within the race for approval in IgAN. As I wrote in my be aware on Vera:
For instance, in November, Japanese Pharma Otsuka Holdings Co., Ltd. (OTCPK:OTSKF) reported that its IgAN candidate sibeprenlimab, after a 12-month Part 2 research, has achieved:
geometric imply ratio discount in 24-hour uPCR from baseline was 47.2%, 58.8%, 62.0%, and 20.0% with sibeprenlimab 2, 4, and eight mg/kg, and placebo, respectively.
This seems to be as aggressive, if not much more spectacular than Alpine’s information taken from a really small variety of sufferers. The Swiss Pharma big Novartis AG (NVS) additionally poses a serious problem to Alpine’s ambitions. Its Part 3 stage candidate atrasentan additionally has delivered some positive Phase 3 data, with the endpoint of lowering ranges of proteinuria being met at 36 weeks, and the corporate planning to push for accelerated approval.
Novartis gained entry to atrasentan through its $3.5bn buyout of Chinook Therapeutics in 2022, and the corporate has two additional late-stage property, in oral complement issue B inhibitor iptacopan, and subcutaneously administered anti-APRIL antibody zigakibart, which has the same MoA to povetacicept and atacicept, and which Novartis believes it could possibly deliver to market by 2027. Approval for atrasentan and iptacopan may happen even sooner, and each have “blockbuster” income expectations.
In the meantime, different corporations creating therapies focusing on IgAN embody BioCryst Prescription drugs, Inc. (BCRX), Ionis Prescription drugs, Inc. (IONS), Omeros Company (OMER), Roche Holding AG (OTCQX:RHHBY), one other Swiss Pharma big, and the generics giants Takeda Pharmaceutical Firm Restricted (TAK) and Viatris Inc. (VTRS).
In brief, alongside not too long ago accredited tarpeyo and filspari, povetacicept should do battle with two medicine with a really comparable MoA, in atacicept and zigakibart, plus quite a few different candidates which might be apparently additional down the road, development-wise. In that context, shopping for Alpine inventory based mostly on Part 2 information from a handful of sufferers might not really feel like a smart transfer.
In such a good race for approval, corporations will want to have the ability to name upon substantial money reserves to fund pivotal research, regulatory issues, R&D, and a possible industrial launch. As of Q3 2023, Alpine reported $197m of present property and a year-to-date web lack of $38m. My guess is that the nearer povetacicept will get to approval, the upper Alpine’s operational prices will climb, and at some stage the corporate will seemingly be compelled to finish additional dilutive fundraisings, negatively affecting its share worth within the course of.
Alpine continues to be led by Govt Chairman and CEO Mitchell Gold, previously CEO of Dendreon Company, the place he helped deliver prostate most cancers immunotherapy Provenge to market, though that drug by no means grew to become the blockbuster asset the corporate believed it may very well be.
Contemplating that Alpine has had two scientific misses, with acazicolcept and davoceticept, it appears important that administration makes probably the most of its alternative with povetacicept, nonetheless, battling the likes of Novartis, and entrenched incumbents equivalent to Travere and Calliditas, is not going to be easy, and it could be value asking the query if Alpina has the industrial nous required to achieve such a crowded house, even when it have been to develop the simplest remedy?
Concluding Ideas – Is Alpine Immune a “Buy,” “Sell” or “Hold” after latest developments?
As talked about above, after neither of its authentic candidates succeeded within the clinic, Alpine is now just about “all-in” on povetacicept.
Clearly, the market has seen one thing it likes within the early povetacicept information readouts, however equally, I discover it tough to search out areas the place the drug is a serious enchancment on present requirements of care, or compared to two different corporations’ medicine with the identical MoA, focusing on the identical illness indications.
As such, I discover a market cap valuation of $2.5bn a little bit excessive for an organization that has thus far arguably encountered extra failure than success, has restricted financing, no prospect of near-term revenues, substantial single asset threat, and loads of competitors.
With that mentioned, I clearly underestimated how a lot the market would recognize information shared by Vera Therapeutics in January, though it ought to be famous that even after its latest rally, it is now value the identical as Alpine, regardless of being additional alongside development-wise.
povetacicept pipeline in a product potential (presentation)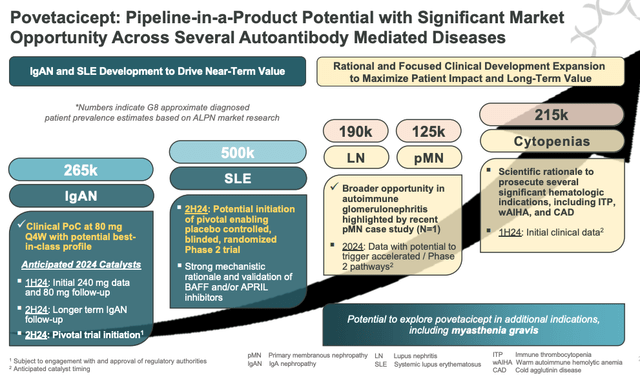

As we will see above, Alpine makes the “pipeline in a product” declare that’s widespread to many biotechs creating a single drug versus a various pipeline, and there are actually causes to consider that povetacicept may efficiently win approvals in all of the markets talked about above – in any case, there are not less than three corporations targeted on creating medicine with the identical MoA, so we should always not underestimate the potential of Alpine’s drug.
With such an extended growth path nonetheless to observe earlier than a industrial approval may be thought of genuinely seemingly, nonetheless, and with no information set massive sufficient to make a bona fide judgment on the standard of Alpine’s product relative to its competitors – of which there’s a lot – whereas I’ll keep away from giving the corporate a “sell” advice, owing to the clear market alternative in play, I will be staying on the sideline in the case of shopping for Alpine inventory.
This area of drug growth, for my cash, is a little bit too dangerous, and I can not level to a transparent benefit that Alpine enjoys over its competitors, not less than presently.