koto_feja
Funding Overview
Ipsen S.A. (OTCPK:IPSEY, IPSEF) is a Paris, France-headquartered Pharmaceutical firm targeted on oncology, uncommon illness and neuroscience. The corporate’s enterprise is constructed upon partnering with different biotech and pharma firms creating new medication. Based on Ipsen’s web site:
Since 2020, Ipsen has introduced in over 20-best- or first-in class applications throughout our pipeline at each stage of improvement and plans are in place to amass extra. We put money into alternatives in difficult illness areas to raised deal with challenges confronted by the neighborhood.
Ipsen gross sales by remedy (Ipsen)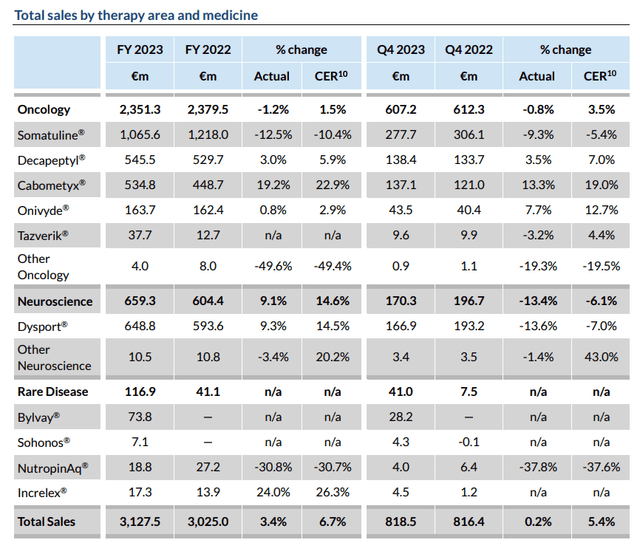

As we will see above, the corporate has a longtime portfolio of business drug product belongings and generated ~$3.4bn of revenues in 2023 – a 3.3% annual uplift. A core consolidated web revenue of ~$830m was reported, versus $946m within the prior yr.
Ipsen’s best-selling merchandise are Somatuline (lanreotide), indicated for administration of acromegaly and signs brought on by neuroendocrine tumors, Dysport, an abobotulinumtoxinA injection indicated for Cervical Dystonia, Facial Wrinkles, Higher Limb Spasticity, and Decrease Limb Spasticity, Cabometyx, a drug it licenses the rights to market and promote outdoors of the US, Canada, and Japan from Exelixis (EXEL), indicated for numerous stable tumor cancers, and Decapeptyl, indicated for prostate most cancers sufferers.
In March 2023, Ipsen acquired Albireo, a “leading innovator in bile-acid modulators to treat pediatric and adult cholestatic liver diseases,” having access to Bylvay, indicated for pruritus and newly authorised to deal with Alagile Syndrome (“ALGS”), which analysts have forecast may obtain “blockbuster” (>$1bn each year) gross sales, particularly if a label enlargement to deal with biliary atresia could be secured.
The corporate says it “anticipates four commercial launches in 2024,” for Onivyde within the indication of pancreatic ductal adenocarcinoma (PDAC), elafibranor in second-line main biliary cholangitis (“PBC”), Bylvay in ALGS, and Sohonos in fibrodysplasia ossificans progressiva (“FOP”), a uncommon musculoskeletal situation.
Onivyde was bought from Merrimack Prescribed drugs in 2017 in a deal price $575m up entrance, plus $450m in potential milestones, whereas Ipsen secured the rights to develop and commercialize elafibranor from Genfit, in a deal price ~$130m up-front, plus as much as ~$390m in milestone funds. Sohonos was acquired by way of Ipsen’s $1bn acquisition of Clementia Prescribed drugs in 2019.
For 2024, administration has guided for a 6% annual income uplift, with core working margin “around 30% of total sales, which includes additional R&D expenses from anticipated early and mid-stage external-innovation opportunities.” That could be a little decrease than prior to now two years, however nonetheless, a powerful margin total.
Long run, administration hopes to drive “total-sales average growth of at least 7% per year for the period 2023-2027 at constant exchange rates” and a “core operating margin in 2027 of at least 32% of total sales.
Ipsen pays a small dividend which yields just over 1%, and is paid in dollars to holders of its ADR stock – the ADR ratio is 1:4 (ORD:ADR). The company reported a cash position of $560m as of the end of 2023, and net cash to debt of ~$70m.
In summary, Ipsen’s relatively unique approach – partnering with or acquiring biotech companies with promising, de-risked assets has been relatively successful to date, although with a current market cap of ~$9.7bn, shares are not especially cheap, and some investments have been better than others – for example, the Clementia acquisition has resulted in a single approval in a small indication, generating only $7m in revenues since its launch in August last year.
As such, and with somatuline’s patent protection having now expired, likely leading to long-term revenue erosion, Ipsen needs its pipeline to deliver some fresh opportunities – which is what makes its newly announced partnership with Sutro Biopharma an intriguing and potentially value accretive deal.
Ipsen Bolsters Pipeline Opportunities With Sutro BioPharma Deal For Antibody Drug Conjugates
Ipsen is at the moment guiding Tazverik – a drug candidate it acquired by way of its buyout of Epizyme in a deal price ~$247m initially, with contingent worth rights hooked up paying $0.3 per CVR “payable upon the primary achievement of $250 million in combination web gross sales of Tazverik” – through a confirmatory study in second line follicular lymphoma treatment, with the drug having been awarded accelerated approval by the FDA in 2020.
A study failure could result in Tazverik being withdrawn from the market, while the drug has not been a commercial success, therefore this study does not add much value to Ipsen’s business.
Ipsen is running Phase 2 studies of two longer-acting neurotoxin drug candidates, and of Fidrisertib, an ALK2 inhibitor licensed from Blueprint Medicines (BPMC), which is also indicated for FOP, as well as running a Phase 2 study of its candidate IPN60250 in Primary sclerosing cholangitis ((PSC)). As shown below, in a slide from a recent investor day presentation, management believes its portfolio is sufficient to drive ~$3bn per annum with no contribution from somatuline.
Ipsen earning potential from selected assets (Ipsen presentation)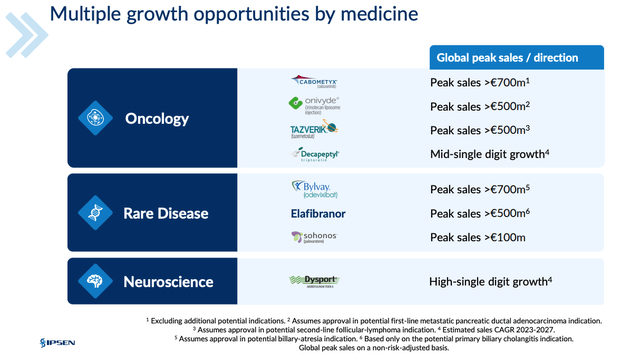

Nevertheless, the pipeline could use one or two more assets of interest, and, as if acknowledging this, last week, Ipsen announced a take care of Sutro Biopharma (STRO), which incorporates:
an exclusive global licensing agreement for STRO-003. STRO-003, an antibody-drug conjugate (“ADC”) in the final stages of pre-clinical development, targets the ROR1 tumor antigen which is known to be overexpressed in many different cancer types including solid tumors and hematological malignancies.1 The agreement gives Ipsen exclusive worldwide rights to develop and commercialize STRO-003 and will be the first ADC candidate joining Ipsen’s expanding portfolio.
Antibody drug conjugates – which combine a monoclonal antibody with a cytotoxic payload connected via a chemical linker, combining the specificity of the antibody with the potency of the payload – are arguably the hottest property in the oncology treatment landscape at this time.
Last year, Pfizer (PFE) agreed to the ~$43bn takeover of Seagen, and its three approved ADCs, Adcetris, Padcev, and Tivdak. AbbVie (ABBV) spent ~$11bn acquiring Immunogen and its folate receptor-alpha-positive ADC Elahere, while Merck (MRK) invested $5.5bn to develop ADCs in partnership with Daiichi Sankyo.
In December last year, Bristol-Myers Squibb (BMY) purchased a Phase 2 stage ADC from China Pharma SystImmune, targeting EGFR and HER3. In January this year, Johnson & Johnson (JNJ) spent ~$2bn acquiring Ambrx, an ADC focused drug developer, and only a couple of days ago, the Danish biotech Genmab (GMAB) agreed to acquire Profound Bio in a $1.8bn deal, having access to:
three clinical and multiple preclinical programs including Rina-S, a potential best-in-class, clinical-stage, FRα-targeted, Topo1 ADC, currently in Phase 2 of a Phase 1/2 clinical trial, for the treatment of ovarian cancer and other FRα-expressing solid tumors.
Ipsen’s deal only gives it access to a single ADC asset – by the terms of the deal:
Sutro Biopharma is eligible to receive up to $900m in potential upfront, development, regulatory and commercial milestone payments including approximately $90m in near-term payments, including an equity investment, and tiered royalties on global sales, contingent upon successful development and commercialization.
Sutro & Ipsen’s Partnership Seems To Suit Both Parties
This feels like a deal that may suit both parties. Ipsen gains access to a promising, albeit early stage ADC, which Sutro says has “proven sturdy monotherapy efficacy and potential for a differentiated security profile in preclinical improvement in stable tumors and hematological malignancies.”
Sutro, for its part, realizes a substantial cash windfall, with the potential for nearly $1bn more in potential payouts, and obtain a powerful Pharma partner which knows how to compete in commercial markets, should STRO-003 progress that far. At the same time, Sutro maintains full control of its lead asset, STRO-002, otherwise known as Luveltamab Tazevibulin, or Luvelta for short.
Sutro is currently enrolling Luvelta in a “registrational” (i.e., data collected from this study may be used to support and approval push, if positive) study in ovarian cancer. Luvelta targets receptor alpha (“FolRα”), similarly to AbbVie’s Elahere, which is also approved to treat advanced ovarian cancer patients, and expected to drive ~$500m revenues in 2024, and potentially, >$2bn in peak annual revenues.
Sutro believes Luvelta may have the edge over Elahere, because it addresses low and high medium expressing FolRα patients, while Elahere is limited to higher expressing FolRα patients only. According to a recent investor presentation, Sutro believes that while Luvelta could potentially serve a patient population of 80% of all of the ADC patient population, Elahere is restricted to 35%.
According to data shared with the American Society of Clinical Oncology, Luvelta achieved an objective response rate (“ORR”) of 43.8% in the higher dose cohort of a Phase 1 study in patients with a FolRα Tumor Proportion Score (“TPS”) of >25%, with a medium duration of response (“mDOR”) of 5.4m, and median Progression Free Survival (“PFS”) of 6.6m.
In its own pivotal study, Elahere was shown to reduce the risk of disease progression or death by 35% versus standard chemotherapy, meeting its primary endpoint, and also a secondary endpoint, showing a 42% ORR across 95 patients, and a 5% complete response rate (11 patients), with mDOR of 6.77m.
As such, the bar for approval seems quite high, however, Elahere’s pivotal SORAYA study was conducted in patients with FolRα TPS >75% (so far as I am aware) and therefore Luvelta may not be required to show superiority or even non-inferiority to Elahere in order to secure approval in a wider patient market. Besides ovarian, Luvelta has achieved an ORR of 29% in 17 patients with FolRα expressing endometrial cancer, and preclinical signs of efficacy in acute myeloid lymphoma (“AML”) and non small cell lung cancer (“NSCLC”).
It is tempting to wonder why Ipsen did not target luvelta as part of its deal for STRO-003 – perhaps Sutro management refused to hand over the rights, figuring it could go it alone – the company reports a current cash position of ~$375m, which it says extends it funding runway to 2H 2025, although a further $75m raise completed last week after the Ipsen deal was made public. Nevertheless, sharing some of the developmental costs for Luvelta might have appealed to Sutro management, although the company has multiple other Big Pharma partners with whom it is sharing its technology – according to its 2023 annual report / 10K submission these include:
an immunostimulatory antibody-drug conjugates collaboration with Astellas Pharma, a cytokine derivatives collaboration with Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc. ((MRK)); a B Cell Maturation Antigen, or BCMA, ADC collaboration with Bristol-Myers Squibb ((BMY)); a MUC1-EGFR ADC collaboration with Merck KGaA, Darmstadt Germany (operating in the United States and Canada under the name “EMD Serono”).
Vaxcyte is an additional partner, leveraging Sutro’s XpressCF® and XpressCF+® platforms, for the “discovery and improvement of vaccines for the therapy and prophylaxis of infectious illness.”
Concluding Thoughts – Ipsen / Sutro Deal May Deliver Long-Term Value For Both Companies – But Sutro Especially Is A Risky Investment
Ipsen seems to have made a smart move by making this deal with Sutro, giving itself access to the potentially very lucrative ADC market. The asset acquired is yet to begin in-human studies, so it seems as though Ipsen is prepared to spend years guiding this asset through the clinical trial process – historically, it has tended to bring in more de-risked assets at later stages of development.
Sutro, for its part, lands a $90m windfall, enabling it to complete its registrational study of Luvelta in ovarian cancer, and fund more studies as a combo therapy in ovarian (alongside chemotherapy), endometrial, NSCLC, and develop a preclinical tissue factor ADC directed against solid tumors.
Sutro’s platform and pipeline has been given additional validation by the number of partners it has attracted – Astellas also paid Sutro $90m upfront, and pledged up to $422.5, in development, regulatory and commercial milestones for each product candidate it develops, while Merck paid $60m upfront in 2018 and is developing a single candidate – MK-1484, a distinct cytokine derivative molecule for the treatment of cancer – with up to $500m milestones on the table.
Investing in either Ipsen or Sutro carries a level of risk, which is substantially higher in the case of Sutro, I would argue. Ipsen’s product portfolio looks capable of maintaining revenues >$3bn per annum for the foreseeable future, and the company and has been long term profitable and dividend paying. The drawback here is the patent expiry of its best-selling asset, and arguably, some slightly over-ambitious peak revenue targets for other portfolio drugs.
From Sutro’s perspective, the company appears to have a strong, late-stage candidate in the form of Luvelta, a decent amount of funding, and, if its technology is validated in the late-stage Luvelta study, for example, or a partner’s, the company may well become an attractive target for M&A, and its share price will likely soar.
There is a tangible risk that Luvelta does not meet the bar for approval in its registrational study, however, which would undermine its technology and eliminate any hope of commercial revenues in the short – to medium term. The ADC drug development market is becoming more crowded and it will be harder for every company to achieve best-in-class status in any indication.
The fact that Sutro’s market cap valuation is just $375m at the time of writing in my view serves as either a warning or an opportunity. Clearly, there is huge upside for shares to explore if Luvelta or one of its partnered drugs makes a clear case for approval – and blockbuster revenues – but partners can walk away from programs without coughing up any milestones, and if Luvelta fails, the company will be back at square one in many ways. Its safety and efficacy must be well-established in the new study, and come very close to matching Elahere, for the company to be certain of an approval shot.
As such, if you are an investor looking to gain some modest exposure to the ADC market, without jeopardizing your investment or risking an overnight >50% loss, then Ipsen S.A. is a company that should definitely be on your radar. If you are looking for a more speculative and potentially rewarding opportunity, Sutro Biopharma, Inc. could be a biotech stock to consider buying.
Editor’s Word: This text discusses a number of securities that don’t commerce on a serious U.S. alternate. Please pay attention to the dangers related to these shares.