vdvornyk/iStock through Getty Pictures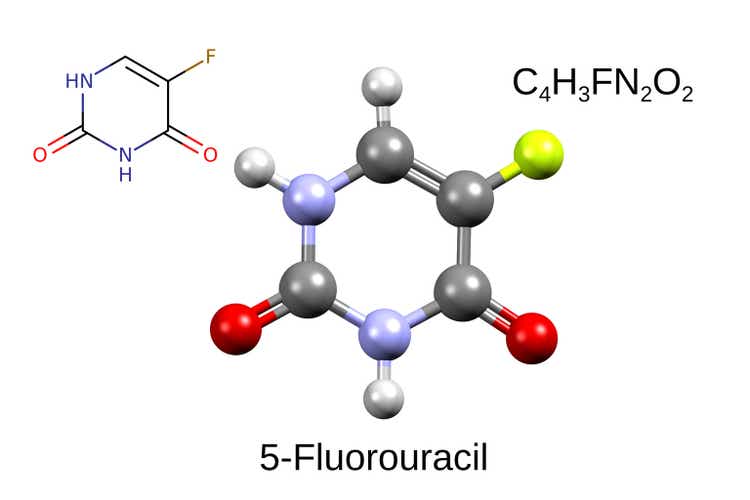

Thesis overview
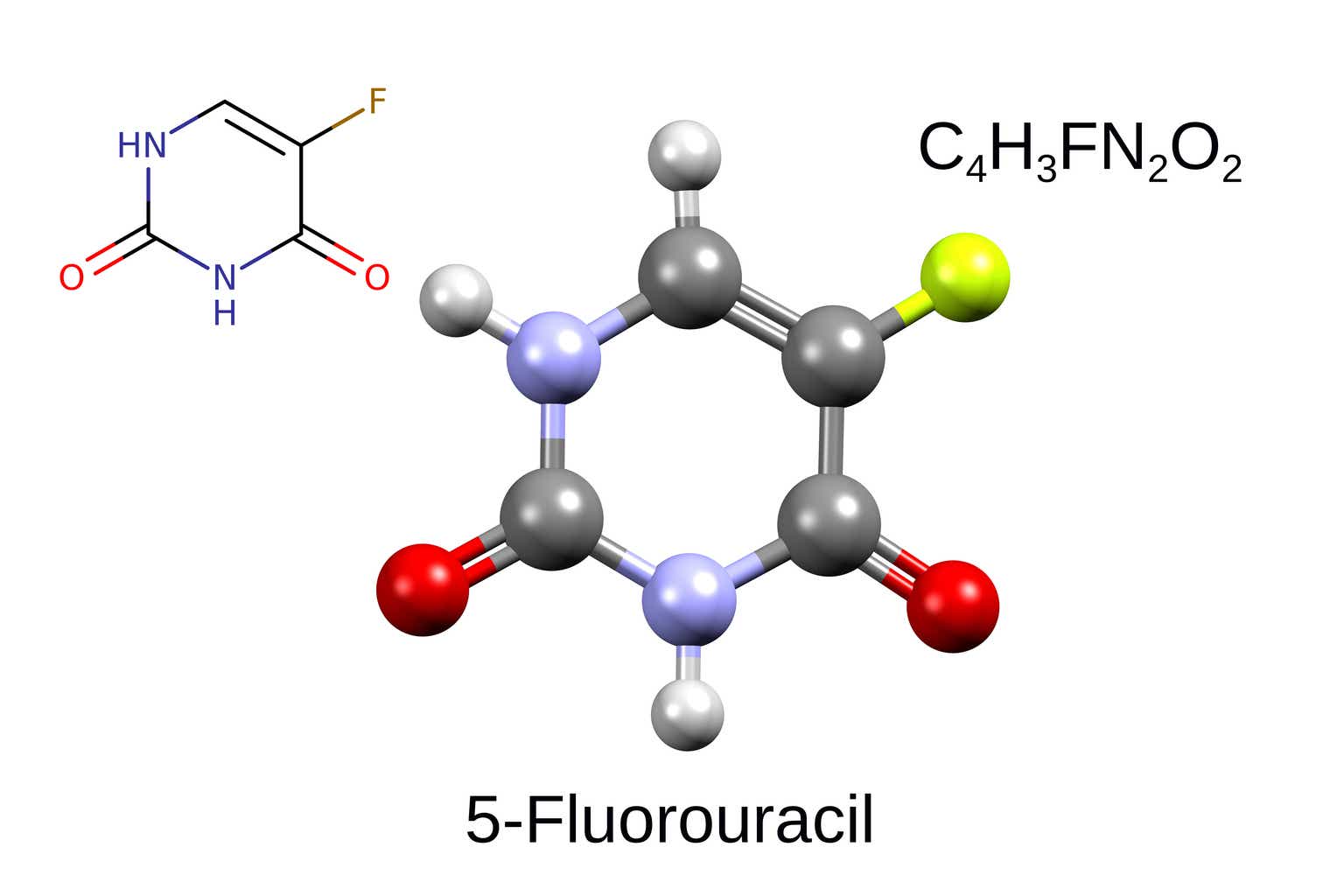
NuCana (NASDAQ:NCNA) is a UK-based biotech growing nucleotide molecules for numerous oncology indications. Its platform, known as ProTide, has main benefits over conventional nucleoside analogues presently used as a part of chemotherapy regimens. NCNA’s lead candidate is NUC-3373 (ProTide 5-fluorouracil). NCNA goals to interchange conventional 5-fluoroucil (5FU) which remains to be getting used as a part of chemotherapy regimens for quite a few malignancies. Importantly, 5FU remains to be used as a part of the spine chemotherapy regimens for 1st-line (1L) and 2nd-line colorectal most cancers (2L-CRC). Preliminary ph1/2 outcomes counsel not solely promising exercise but in addition higher security and tolerability. An ongoing randomized managed trial (RCT) in 2L-CRC is recruiting properly, with full enrolment anticipated inside the coming months and a readout probably in 2024. A constructive readout, which for my part is very probably, ought to end in vital upside and open alternatives for funding/partnerships. Though my thesis relies on this binary catalyst, there are a number of further readouts from ongoing ph1/2 trials anticipated in 2024.
The principle danger to the thesis is an underwhelming readout in above-mentioned RCT. Further dangers embrace want to boost money someday in 2024, in addition to danger of reverse break up by Could 2024.
Why is the inventory down?
On the time of writing NCNA is buying and selling at a 90% low cost in comparison with March 2022. There are a number of causes for the downward pattern since then;
- In March 2022 NCNA introduced discontinuation of a part 3 examine of its then lead candidate, NUC-1031 (ProTide gemcitabine), after an interim futility evaluation. Notably, NUC-1031 remedy arm did worse (decrease survival) than the management arm. Earlier than that, NUC-1031 had failed one other part 3, in pancreatic most cancers (though failure was attributed to unfavorable baseline imbalances in affected person traits, and constructive survival traits had been noticed in chosen affected person sub-groups). These failures had been main hits for NCNA placing into query the entire oncology ProTide platform. Failure of NUC-1031 and implications for the remainder of the pipeline can be mentioned within the subsequent part. Notably, NCNA was buying and selling at $2.9 earlier than discontinuing the ph3 in biliary tract cancers, and at round $7 earlier than failure of the ph3 in pancreatic most cancers.
- A number of readouts from ongoing ph1/2 research had been eagerly anticipated by traders throughout 2024. Nevertheless, readouts apparently didn’t meet traders’ expectations (to be mentioned in additional element in a subsequent part). Importantly, traders had been in all probability anticipating an interim readout from ongoing part 2 RCT in 2L CRC in 2023. Nevertheless, as a substitute of that we obtained an abstract describing the examine design, with no efficacy knowledge. The one new data was that the “examine is recruiting properly and aggregated security knowledge from the primary 40 patients enrolled confirmed no new security alerts”.
- NCNA guides a money runway to 2025. Subsequently, much more money can be wanted to advance pipeline to later-stage scientific trials, which with no partnership can be severely dilutive contemplating present market cap ($15.6M on the time of writing). Notably, the guided runway relies on elements which might be past NCNA’s management (to be mentioned within the “Financials” part).
- Danger of de-listing or reverse break up by Could 2024. NCNA first acquired delisting discover in May 2023. In November 2023 NCNA announced itemizing switch to Nasdaq Capital Market and an extra 180-day interval (i.e as much as Could 6, 2024) to regain compliance (bid worth>$1).
- Throughout early 2023 inventory was buying and selling at $1.5-1.6 attributable to hopes of successful a patent litigation in UK and Germany in opposition to Gilead for merchandise containing antiviral sofosbuvir (examine coverages from E. Roudasev for extra particulars on this). Nevertheless, Gilead received, regardless of a previous favorable choice in Germany. Following adverse selections (by the Patents Court docket of the Excessive Court docket of Justice of England and Wales, and by EPO Technical Board of Enchantment) that invalidated the related patents, NCNA has decided “to abandon further proceedings in Germany” and have become “liable to pay a proportion of Gilead’s legal fees” for authorized proceedings in UK and Germany. “All obligations arising from the patent infringement litigation in the U.K. and Germany have been settled as of September 30, 2023.”
Overview of ProTide platform
Nucleoside analogues are nonetheless broadly used as a part of chemotherapy regimens however have main limitations;
- Fast breakdown leads to brief half-life, which necessitates difficult administration schedules (e.g. 5FU is usually administered as a 46-hour steady infusion). Moreover, breakdown into poisonous metabolites (as is the case for 5FU) is accountable for lots of the noticed antagonistic results ((AEs)).
- Their uptake by cells requires lively transport. This, together with fast breakdown, leads to poor intracellular focus (the place the drug works), particularly when the required transporters are usually not expressed at enough stage by tumor cells.
- Nucleoside analogues have to be transformed intracellularly to the lively substances by way of a posh metabolic pathway. Expression of the required enzymes fluctuate from individual to individual and could also be low in most cancers cells, thus leading to unpredictable (and infrequently inadequate) activation to the lively metabolite. Moreover, this course of leads to era of poisonous metabolites.
ProTide know-how avoids above limitations, leading to nucleotide analogues which might be protected against breakdown, can higher penetrate cells (no want for lively transport) and are usually not depending on complicated intracellular enzymatic activation. In consequence, larger intracellular focus of the lively metabolite will be achieved (which can translate to higher efficacy), whereas at identical time avoiding era of poisonous metabolites (which can translate to higher security/tolerability profile). Furthermore, higher pharmacokinetic traits permit simpler administration schedules (e.g. 2h infusion for NUC-3373 vs 46h infusion for conventional 5FU).
Limitations of conventional nucleoside analogues and benefits of the ProTide platform are summarized within the picture under;
Shortcomings of nucleoside analogs and the ProTide resolution (2023 Annual Report)

Following failure of NUC-1031 (ProTide gemcitabine), the present lead ProTide candidate is NUC-3373 (ProTide 5FU). Moreover, NCNA is growing NUC-7738 (ProTide 3′-deoxyadenosine), which nevertheless remains to be in early phases of scientific improvement.
NCNA pipeline (Firm presentation)

Evaluation of NUC-1031 failure and implications
As mentioned above, NUC-1031 (Acelarin), NCNA’s prior lead candidate, has failed two part 3 trials (in biliary tract most cancers and in pancreatic most cancers), regardless of promising earlier stage knowledge. A abstract of scientific trial outcomes of NUC-1031 (together with in different indications) will be discovered within the newest annual report. NUC-1031 will not be the explanation why I’m invested in NCNA, nevertheless for a lot of it has been a serious motive to cross on an funding. Within the dialogue under I’ll clarify why failure of NUC-1031 will not be predictive of failure of NUC-3373.
To summarize what can be mentioned in additional element under, though failure of Acelarin in two part 3 trials is regarding in regards to the ProTide platform, NUC-3373 is a special molecule, being developed for a special indication (CRC), with higher security/tolerability profile in comparison with Acelarin and promising efficacy alerts (to be mentioned in a subsequent part) in scientific trials. Moreover, failure of Acelarin in biliary tract most cancers has been impacted by a excessive early discontinuation fee and pitfalls within the protocol (remedy cessation as a substitute of re-challenge at a decrease dose). Such issues don’t apply to NUC-3373 primarily based on thus far obtainable knowledge. Subsequently, there may be little motive to consider that NUC-1031 failure is predictive of NUC-3373 failure. Quite the opposite, there are legitimate causes (to be mentioned in subsequent sections) to anticipate a profitable scientific improvement of NUC-3373.
If in case you have restricted time simply skip subsequent paragraphs and transfer on to the following part.
Acelarin was superior straight to a part 3 trial in biliary tract most cancers following a part 1b examine. The phase 1b was an open-label, single-arm, dose‐escalation examine that enrolled n=21 sufferers with first-line (1L) superior biliary tract most cancers. Therapy was reported as “well tolerated”. Nevertheless, on the dose used within the subsequent phase 3 trial (NuTide:121), Grade 3 AEs γ-GT elevation was famous in 67% of the sufferers. Moreover, 9 of 21 sufferers (43%) discontinued the remedy attributable to antagonistic occasions. As can be mentioned under, NUC-3373 has a significantly better security profile. I’m emphasizing this as a result of failure of Acelarin has been impacted by a excessive early discontinuation fee attributable to AEs, a difficulty that doesn’t apply to NUC-3373.
To my data outcomes of the part 3 trial have solely been revealed within the type of an abstract. So detailed outcomes are usually not obtainable. Primarily based on obtainable knowledge NUC-1031 handled sufferers had the next goal response fee (18.7% vs 12.4%). Nevertheless, median general survival (9.2 vs 12.6 months) and PFS (4.9 vs 6.4 months) had been decrease. Subsequently, not solely did NUC-1031 fail to beat commonplace of care, however even carried out worse.
Grade ≥ 3 AEs had been related between the two teams with the exception of: “liver-related events [increased ALT (17.8% vs 3.4%) and AST (9.1% vs 2.4%), both higher in NUC-1031+cis], hepatobiliary disorders [cholangitis (3.4% vs 1.6%) and biliary obstruction (1.8% vs 0.3%), both higher in NUC-1031+cis]; and haematological [anaemia (9.4% vs 18.0%) and neutropenia (14.1% vs 24.1%), both higher in GemCis].” Extra detailed knowledge are usually not obtainable, however one might hypothesize that the upper fee of hepatobiliary AEs could also be related to stronger tumor responses (= probably stronger native irritation damaging liver tissue, contemplating that 54% of sufferers had intra-hepatic tumors). Notably, regardless of related Grade ≥ 3 AEs evaluating the two arms, discontinuation fee attributable to AEs was a lot larger within the NUC-1031 arm (30 vs 16%), leading to considerably lower treatment exposure (“median number of cycles delivered of four vs six in the control arm”). Moreover, early discontinuation (inside 30 days) was extra frequent in NUC-1031 arm (22% vs 10%, elsewhere this has been reported as 11% vs 1%, or 14.6% vs 2.6%), and had been predominantly attributable to Grade 3 ALT/AST will increase, which had been reversible. Nevertheless, the protocol was amended (permitting re-challenge at a decrease dose) solely late through the trial progress. The above might clarify the discordance between ORR (higher with NUC-1031) and PFS/OS (worse with NUC-1031).
Of be aware can be that “NuCana believes that the pattern of liver-related TEAEs observed in NuTide:121 is specific to biliary tract cancer and potentially the combination with cisplatin. The pattern of liver-related TEAEs observed in NuTide:121 was not observed in trials that assessed Acelarin as a monotherapy (PRO-001, PRO-105 and ACELARATE) or in combination with carboplatin (PRO-002). Furthermore, this pattern of liver-related TEAEs has not been observed in clinical studies assessing NUC-3373 as a monotherapy (NuTide:301) or in combination with leucovorin, bevacizumab and either irinotecan or oxaliplatin (NuTide:302) or in the clinical study assessing NUC-7738 as a monotherapy (NuTide:701)”. In different phrases, liver toxicity noticed with NUC-1031 in NuTide:121 doesn’t characterize a platform (ProTide)-wide danger. Most, probably it would not even apply to different malignancies, and should even be a limitation of the particular chemotherapy routine used.
Primarily based on the above, there are legitimate causes to consider that failure of the part 3 was attributable to extreme pitfalls within the trial design. In my view, I would not exclude the chance that Acelarin is healthier than commonplace gemcitabine (contemplating larger ORR). Excessive early discontinuation due hepatic negative effects led to a lot shorter remedy period and in consequence much less sturdy responses (therefore decrease PFS and OS). A extra appropriately designed trial, permitting dose reductions (quite that remedy cessation) in case of liver toxicity might have resulted in higher efficacy.
Contemplating the above, NCNA has not utterly deserted Acelarin and is “assessing future development options for Acelarin in biliary tract cancer which may explore lower doses of Acelarin, alternative combination partners or specific sub-sets of biliary tract cancer patients. Indications other than biliary tract cancer are also being assessed as future development options for Acelarin”. Clearly Acelarin scientific improvement will not be a precedence for NCNA, and my thesis will not be primarily based on Acelarin potential. However I would not exclude profitable eventual improvement of Acelarin a couple of years from now, particularly after additional proof-of-concept is established by ongoing trials.
Overview of NUC-3373
As mentioned above, NUC-3373 (ProTide 5FU) goals to deal with shortcomings of conventional 5FU. Preclinical knowledge, in addition to early scientific knowledge counsel potential for improved efficacy, in addition to improved tolerability and security. Importantly, NUC-3373 is being administered as a 2-hour infusion (in comparison with the 46-hour infusion by a conveyable pump of 5FU).
I’m not going to deal with the preclinical knowledge right here (I refer readers to the newest annual report) however it’s value mentioning that NUC-3373 has been proven to be far more potent that 5FU in opposition to quite a lot of most cancers cell traces (together with CRC, ovarian and lung most cancers) and should have further anti-cancer mechanisms of motion (together with induction of immunogenic cell loss of life). The latter implies that NUC-3373 might enhance efficacy of immunotherapies.
Close to scientific knowledge, NUC-3373 is being evaluated in 3 trials;
- Two trials in colorectal most cancers; NuTide:302 (ph1b/2 in superior CRC) and NuTide:323 (ph2 RCT in 2L CRC). That is presently the lead program and for good causes. 5FU stays the cornerstone of CRC remedy regimens in a number of traces (together with 1st and 2nd line). Notably, “approximately 500,000 patients in North America are estimated to receive intravenous 5-FU each year”. Simply contemplating CRC, there are about 150K new instances identified per yr simply within the US. Subsequently, simply CRC can be an enormous goal marketplace for NCNA.
- NuTide:303 is a ph1b/2 evaluating NUC-3373 in different strong tumors, to be able to determine potential targets past CRC. In NuTide:303 NUC-3373 is being evaluated together with pembrolizumab (PD-1 inhibitor) “for patients with advanced solid tumors and in combination with docetaxel for patients with lung cancer”.
5FU stays the cornerstone of CRC (Firm presentation)

NuTide:302
NuTide:302 is a 3-part trial;
- Half 1; Sufferers with superior CRC which have failed at the least 2 prior 5-FU-based regimens (i.e. 3L+, median of 4 prior chemotherapy regimens). The purpose of Half 1 was to find out whether or not NUV-3373 must be administered with leucovorin. Primarily based on Half 1 it was established that leucovorin has no impression on the pharmacokinetic or security profile of NUC-3373.
- Half 2; Sufferers with superior CRC which have failed at the least 2 prior 5-FU-based regimens (i.e. 3L+, median of three and 4 prior chemotherapy regimens in NUFOX and NUFIRI arm, respectively). Half 2 was a dose escalation examine of NUC-3373 with irinotecan (NUFIRI) or oxaliplatin (NUFOX) to search out the Really helpful Section II Dose (RP2D).
- Half 3 (ongoing) is assessing NUFIRI/NUFOX + bevacizumab in 2L CRC sufferers (1 prior line of 5FU-based chemotherapy).
Each Half 1 and a couple of have been efficiently accomplished. Each half 1 and a couple of, in addition to interim outcomes of half 3, have proven a good security/tolerability profile (higher than anticipated by conventional 5FU, no Grade 4/5 AEs) in addition to encouraging indicators of efficacy. Of be aware are the next; (1) Responses (partial response/secure illness) had been noticed in closely pre-treated sufferers which have failed at the least 2 prior 5FU-based regimens (most 4L+ in Elements 1 and a couple of). (2) Length of responses (DOR) was in lots of sufferers longer than in prior (sometimes DOR is predicted to get progressively shorter shifting to later traces of remedy).
Abstract of security knowledge from NuTIde:301 and NuTide:302. Be aware low fee of Grade 3-4 AEs, in comparison with 5FU and capecitabine (Extracted from firm presentation) Efficacy knowledge displaying longer than anticipated period of responses (Firm presentation)



NuTide:323
NuTide:323 is a part 2 RCT in 2L CRC. Because the comparator arm is FOLFIRI+bevacizumab, the trial will enroll sufferers for which bevacizumab (quite than focused therapies) is suitable. One of many targets of the trial is to match a Q1W vs Q2W NUFIRI+bevacizumab routine.
Design of NuTideL:323 (Firm presentation)

NUFIRI is being administered on the most tolerated dose as outlined in Half 2 of NuTide-302 (NUC-3373 1500 mg/m2 + LV 400mg/m2 + irinotecan 180 mg/m2), which as depicted above was related to few Grade 3 AEs and no Grade 4 AEs. Notably, this NUC-3373 dose is decrease than the utmost tolerated dose of 2500 mg/m2 weekly, as decided in an earlier part 1 trial, Nutide-301(this contrasts Acelarin improvement the place the very best dose was superior within the part 3, leading to early remedy discontinuation and shorter period of response regardless of larger goal response fee).
NCNA goals to point out superiority of NUC-3373 over 5FU, which might be nice. Nevertheless, for my part, even non-inferiority (i.e. NUC-3373 being at the least nearly as good as 5FU by way of scientific efficacy) would additionally make a compelling case in favor of NUC-3373 contemplating higher security/tolerability profile, in addition to affected person comfort (NUC-3373 is being administered over a 2-hour infusion, whereas 5FU is being administered as a steady 48h-infusion, i.e. the affected person has to hold an infusion pump for two days).
NuTide-323 is enrolling solely a selected subgroup of 2L CRC sufferers (for which bevacizumab quite than different therapies is suitable). Nevertheless, if the trial is succesful it’s cheap to anticipate that NUC-3373 ought to be capable of exchange 5FU in different 2L+ CRC sufferers, in addition to in 1L CRC sufferers (and doubtless in further oncology indications the place 5FU remains to be getting used). Subsequently, a constructive readout (not essentially superiority, as mentioned above) ought to end in main upside in NCNA inventory worth.
When to anticipate a readout for NuTide:323?
Based on the newest PR (Oct 2023) 40 sufferers had been enrolled by that point. Contemplating that the first affected person was enrolled in 18 April 2023, this corresponds to about 6-7 sufferers per 30 days, which is sluggish contemplating money runway. Nonetheless, it is not uncommon for enrolment in scientific trials to progress a lot quicker because the examine progresses and extra websites are initiated (trial is presently being performed in 55 sites in keeping with ClinicalTrials.gov; 15 web site in US, 9 websites in France, 4 websites in Germany, 6 websites in Italy, 14 websites in Spain, and seven websites in UK). Based on NCNA examine stays on track for full enrolment “in the coming months” (in keeping with ClinicalTrials.gov estimated main completion date is June 2024, whereas estimated examine completion date is December 2024) and NCNA expects to report “data” (hopefully efficacy knowledge) in 2024.
Within the part 3 trial of Acelarin NCNA efficiently enrolled n=761 sufferers with biliary tract most cancers from Dec-2019 to Mar-2022 (i.e. over 27 months, equivalent to a mean enrolment fee of 28 sufferers per 30 days). That examine had been performed in additional than twice the examine websites (125 websites throughout 15 international locations), however incidence of biliary tract most cancers (about 8,000 sufferers in US per yr) is way decrease in comparison with incidence of CRC (about 150,000 sufferers in US per yr). Subsequently, I’m assured that NCNA will meet above timelines for NuTide:323 and report a readout someday in 2H 2024 (though an earlier interim readout is theoretically doable).
NUC-7738
NUC-7738 is a ProTide transformation of cordysepin, a nucleoside analogue that has failed scientific improvement attributable to above-discussed limitations of nucleoside analogues (predominantly the fast breakdown). NUC-7738 is being evaluated within the ph1/2 NuTide:701 trial. Section 1 half (a dose escalation examine of NUC-7738 monotherapy) has been accomplished, whereas part 2 (both as monotherapy or together with pembrolizumab) is ongoing. My thesis will not be primarily based on NUC-7738m however it’s value mentioning that encouraging efficacy alerts have been reported thus far in quite a lot of tumors, with a good security profile.
For instance, NCNA lately reported preliminary outcomes of NUC-7738 together with pembrolizumab ((anti-PD-1)) in sufferers with superior/ metastatic cutaneous melanoma who had progressed on 1- 2 prior traces. Notably, responses had been noticed in sufferers that had progressed after prior anti-PD-1 primarily based remedy, suggesting that NUC-7738 might potentiate exercise of anti-PD-1.
Design of the ph1/2 examine for NUC-7738. Section 2 half is ongoing. (Firm presentation) Security profile of NUC-7738 (Firm presentation) Outcomes from ongoing ph2 half (Firm presentation)





Financials
NCNA reported money and money equivalents of £17.8M as of Sep 2023. Including to that £9.43M present revenue tax receivable and £3.41M “prepayments, accrued income and other receivables” the overall sum is £30.64M. For the 9 months ended Sep 2023 working bills had been £22.98M (R&D £18.2M and administrative bills £4.78M), equivalent to money burn fee of £2.55M per 30 days. At this money burn fee runway must be about 12 months (30.64/2.55), i.e. as much as Sep 2024, which must be sufficient to final by way of main catalysts (primarily readout of RCT in 2L CRC).
Above money runway estimation is shorter than NCNA’s guidance of an “anticipated runway into 2025”. It is because NCNA expects a decrease money burn fee going ahead as above-mentioned 9-month money burn has “been impacted by the payment of accruals for clinical trial expenses relating to the Phase 3 clinical trial of Acelarin and the settlement of obligations arising from the patent infringement litigation in the U.K. and Germany”. R&D bills for Acelarin throughout that interval had been £1.93M, whereas prices arising from the patent infringement litigation associated to Gilead litigation was £3M. Subsequently, subtracting these 2 prices (£1.93M+£3M) would end in discount of month-to-month money burn by £0.55M. Contemplating a diminished money burn of £2M ( £2.55M minus £0.55M) estimated runway is prolonged to fifteen.3 months (30.64/2), i.e. to early January 2025 (which matches NCNA’s guided runway).
Of be aware, guided runway relies on tax receivables, the timing of which is unsure (“We plan to fund our cash flow needs through current cash on hand together with the receipt of research and development tax credits from HMRC, the timing of which is outside of our control“). Taking tax receivables out of the equation and contemplating £17.8M money and £2M month-to-month working bills would leads to a a lot shorter runway of about 9 months (i.e. June 2024). Subsequently, anybody investing in NCNA now ought to pay attention to the danger of dilutive money increase someday in 2024 (“In assessing the requirements necessary to continue progressing our research and development activities as currently anticipated and considering the material uncertainty regarding the timing of future cash inflows from research and development tax credits relating to the year ended December 31, 2023, there will be a requirement to seek additional capital to fund operations”). Hopefully, this can be after a constructive readout, however elevating money earlier than any main catalyst is probably going.
Dangers
Anybody contemplating an funding in NCNA ought to pay attention to the next main dangers;
- A very powerful danger to the thesis is a adverse readout from the continued RCT in 2L-CRC. Though I predict a constructive end result for the explanations mentioned above, a adverse end result can be catastrophic for shareholders and for ProTide platform.
- Until there’s a main constructive readout earlier than Could 2024, chances are high that NCNA will not be capable of meet continued itemizing necessities (particularly a bid worth > 1$). Subsequently, almost certainly it’s best to anticipate a reverse break up by Could 2024. There may be additionally the theoretical danger of de-listing, if a call is made in opposition to a reverse break up.
- As mentioned above NCNA’s money runway is a serious concern and a dilutive money increase must be anticipated someday in 2024. Nonetheless, I consider that constructive outcomes from ongoing trials, particularly the RCT in 2L CRC, will open alternatives for funding and/or partnerships. Then again, there may be the danger that NCNA may elect to boost money earlier than pending readouts, which might almost certainly be dilutive.
Notably, NCNA is rated as a “strong sell” by In search of Alpha’s quant system, with low grades in all elements (development, profitability, momentum, revisions) besides valuation. This displays the excessive danger related to investing on this inventory.
Conclusion
NCNA is a beaten-down inventory and for good causes, together with failure of prior lead candidate in a part 3 trial (placing the entire ProTide platform into query), precarious monetary scenario, and danger of delisting. Nevertheless, there are legitimate causes to consider that present lead candidate will succeed, which is able to open a big goal marketplace for NCNA and may end in main revenue for traders. Contemplating delisting / reverse-split danger by Could 2024, it’s doable that the downtrend might proceed pending scientific trial readouts. Subsequently, it might be safer to re-consider investing in NCNA someday later in 2024, doable after a reverse break up and earlier than readout of the continued RCT. For me, the uneven danger/reward is value a wager and I consider the inventory has been punished sufficient in 2023 (tax-loss harvesting might need additionally performed a task within the downward pattern of the inventory the final couple months). Subsequently, I’ve began a small place, and should add later in 2024 (relying on additional information by then). However I’ve to focus on once more this isn’t an funding for everybody and there may be appreciable draw back danger.
Your suggestions is appreciated
Please remark under in case you have any suggestions (constructive or adverse), in case you spot any errors, or in case you consider I missed one thing vital in my evaluation.
Additionally I counsel monitoring feedback if you’re eager about following the inventory as I could put up updates there.
Editor’s Be aware: This text covers a number of microcap shares. Please pay attention to the dangers related to these shares.